Plastic Surgery News: Behind Gummy Bear Implants & Acellular Dermis Matrices

Plastic and cosmetic surgery has gone through many innovations. Many implants and techniques for breast augmentation surgeries utilize technology and procedures that simply weren’t available before.
Today’s news highlights will be gummy bear implants as well as Acellular Dermal Matrices (ADM).
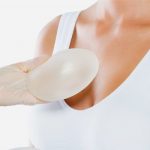
Gummy Bear Implants
The Food and Drug Administration (FDA) approved form-stable silicone gel breast implants in March 2012. It took 8 years of clinical trials by the medical aesthetics company Sientra to ensure that their new implants were safe and effective.
Its nickname – gummy bear implants – was first coined by Californian plastic surgeon Dr. Grant Stevens, who noted its similarities to gummy bear candies. Both the outer shell and the contents within are made from the same materials – in this case, cohesive silicone gel. But while some see similar properties between the implant and the popular snack, the FDA and Sientra themselves discourage such a label. In an interview with ABC News, the company’s CEO Hani Zeini expressed that comparing the ‘medical device to a piece of candy trivializes it’.

There’s also less wrinkling or rippling, which is a common problem especially when smooth silicone gel implants are used. Patients have cited additional firmness, especially for those who chose gummy bears for their secondary procedures. Because it is textured, the implant lowers the risk of capsular contracture or the formation of hardened scar tissue around the implant.
Just like any medical device, this type of implant also comes with its own issues. Should it rotate, its irregular shape may cause a more noticeable distortion than its round counterpart. A longer incision is also necessary to ensure proper placement either above or below the pectoral muscle. Though rare, there are still cases of infection, bleeding, and rupture post-surgery. According to the American Society of Plastic Surgeons, however, this particular variety is ‘thought to be less likely to break.’
To date, there are three licensed manufacturers and distributors of the implants: Sientra, Allergan, and Mentor.
Acellular Dermis Matrices
The extra skin within the breast eliminates the need for weeks-long expansion of the pectoral muscle and breast skin to create a pocket suitable size for the implant. This is often the case with breast cancer patients who had some or all of their breast tissue removed in a mastectomy and want immediate reconstruction. When patches of ADM are stitched along the muscle or on the underside of the breast, there is also better coverage for implant edges.
Surgeons have reported that “rapid early expansion (using acellular dermis) has led to improved cosmetic outcomes,” as cited in Medscape. Also, despite the matrix being foreign to the chest area, there are no issues regarding the repopulation and revascularization of the implanted tissue. Patients are assured of healthy integration of their cells into the matrix.
Among the most preferred matrix options are AlloDerm, DermaMatrix, and Tutoplast which are donated human tissue, and Strattice which is sourced from pigskin. Before cosmetic use, ADM was utilized in burn and abdominal wall reconstructions.
Continuous Innovation is Key
These are just two of the many, many new procedures developed for plastic and cosmetic surgery. They are all results of years of rigorous testing, designed to give you only the very best results. Do not be afraid to ask about them when considering plastic surgery.
*This blog is created and maintained for informational purposes only. The images present may not accurately reflect actual cases per individual. Individual cases are unique, and the descriptions and solutions will vary per patient.